Internal Consistency Analysis
An independent approach for evaluating the accuracy of data is the examination of the internal consistency of the CO2 system parameters. The CO2 system in seawater can be defined by knowing T, S, nutrient and boron concentrations, and two of the four measurable carbon parameters: TCO2, TALK, fCO2, and pH. Thus, cruises on which three or more carbon parameters were measured were overdetermined. By comparing the carbon system distributions estimated from different pairs of carbon measurements on a cruise one can evaluate potential offsets in one of the measured parameters. By examining the internal consistency over several cruises, one can gain confidence in the reliability of the constants being used and evaluate the implied offsets.
The constants of Mehrbach et al. (1973) as refit by Dickson and Millero (1987) were used for this analysis, along with equilibrium constants for other components (e.g., boric acid dissociation, solubility of CO2, water hydrolysis, and phosphoric and silicic acid dissociation) necessary to characterize the carbonate system in seawater as recommended in Millero (1995). The selection of the constants of Mehrbach et al. (1973) was made based on the analysis of a large field data set (15,300 samples) obtained from all the ocean basins (Lee et al. 2000).
This approach relied heavily on two basic premises. The first was that all of the pH measurements needed to be adjusted upward by 0.0047. The justification for this adjustment is discussed in detail in the Adjustments Page. The second premise was that offsets in the internal consistency checks were attributed to errors in the TALK measurements. This premise is supported by the crossover analysis and other approaches which suggested that the other carbon parameters were within acceptable ranges for the cruises in question.
We compared measured alkalinity vs. alkalinity calculated from two different input combinations: pH+TCO2 or fCO2+TCO2.
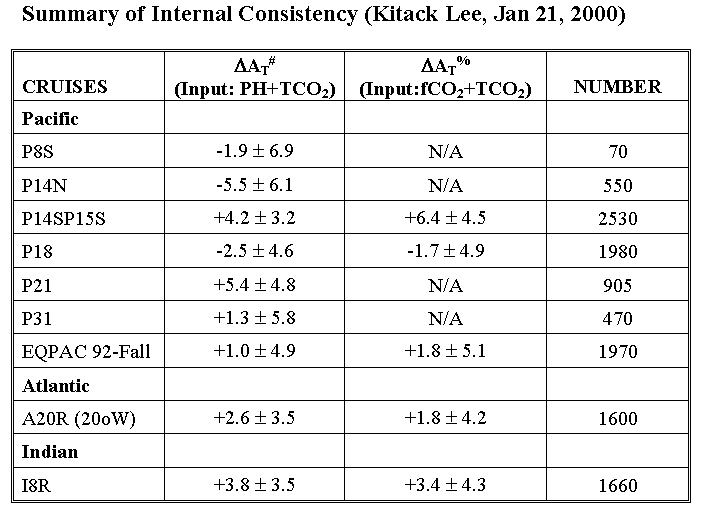
# ΔAT
(µmol/kg) = (measured - calculated). pH data adjusted
upward by 0.0047 were used in these calculations. Without pH adjustment,
+5 µmol/kg should be added to ΔAT.
% ΔAT (µmol/kg) = (measured - calculated).
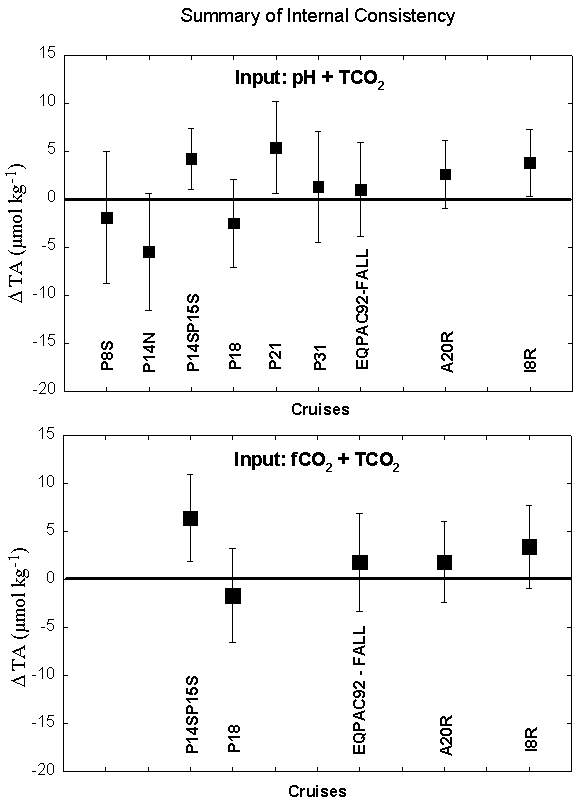
This analysis showed that the measured values of TALK for section P14N are approximately 5 µmol/kg lower than the TALK calculated from TCO2 and pH. Section P21 and P14S15S had measured values that were higher than the calculated values by 5 µmol/kg (based on TCO2/pH pair) and 4-6 µmol/kg (based on TCO2/pH and TCO2/fCO2 pairs), respectively. Note that proposed TALK adjustments implicitly assume that pH, fCO2, and TCO2 data are internally consistent from cruise to cruise. All of these results must be considered in concert with the other lines of evidence on the quality of the TALK measurements from the various cruises. The TALK data were also examined using a basin-wide MLR approach, with crossover calculations, and using an isopycnal analysis. There is a summary tablea> of the TALK quality assessment results, as well as an Adjustments Page with a synopsis of the adjustments we propose for the Pacific dataset.
References
- Dickson, A.G., and F.J. Millero. 1987. A comparison of the equilibrium constants for the dissociation of carbonic acid in seawater media. Deep-Sea Research 34:1733-43.
- Lee, K., F. J. Millero, R. H. Byrne, R. A. Feely and R. Wanninkhof. 2000. The Recommended Dissociation Constants of Carbonic Acid for Use in Seawater. Geophysical Research Letters 27:229-32.
- Merhbach, C., C.H. Culberson, J.E. Hawley, and R.M. Pytkowicz. 1973. Measurement of the apparent dissociation constants of carbonic acid in seawater at atmospheric pressure. Limnology and Oceanography, 18:897-907.
- Millero, F.J. 1995. Thermodynamics of the carbon dioxide system in the oceans. Geochim. Cosmochim. Acta, 59:661-77.
